Nitrate Removal Using Electrokinetic/Iron Wall Processes
C.F. Chew and T.C .Zhang
125B Engineering Hall, Dept. of Civil Engineering, University of Nebraska-Lincoln at Omaha Campus, Omaha, NE 68182-0178, Phone: (402)554-3784; Fax: (402)554-3288
Abstract
The feasibility of using electrokinetics coupled with a zero-valent iron (Fe0) treatment wall to abiotically remediate nitrate-contaminated soils was investigated. Upon completion of each test run, the contaminated soil specimen was sliced into five parts and analyzed for nitrate-nitrogen, ammonia-nitrogen, and nitrite-nitrogen. Nitrogen mass balance was used to determine the major transformation products. In control experiments where only electrokinetics was used at various constant voltages, 25 to 37% of the nitrate-nitrogen was transformed. The amount of nitrate-nitrogen transformed improved when a Fe0 wall (20 g or about 8-10% by weight) was placed near the anode. For test runs at various constant voltages, the amount of nitrate-nitrogen transformed ranged from 54 to 87%. By switching to constant currents, the amount of nitrate-nitrogen transformed was about 84 - 88%. The major transformation products were ammonia-nitrogen and nitrogen gases. Nitrite-nitrogen was around 1% or less in all experimental runs.
Keywords: electrokinetics, iron wall, zero-valent iron, nitrates, groundwater, remediation
Introduction
The prevalence of nitrate contamination in groundwater and the lack of inexpensive in situ methods to treat the contamination is a problem of great concern to the United States. In Nebraska alone, nitrate concentration exceeded the maximum contamination level (MCL) in more than 20% of the 5,286 wells sampled between 1984 and 1988 (Exner and Spalding, 1990). Within the contaminated area, the average nitrate-nitrogen increased at a rate of 0.4 to 1.0 mg/L per year to average concentrations of 1.5 to 2.0 times the MCL (Exner and Spalding, 1991). Treatment methods such as in situ bioremediation or in situ chemical treatment face the problems of moving nutrients, inoculated bacteria, and treatment chemicals through soil to the proximity of contaminants using hydraulic pressure. Because of this, there is a need for technologies to remediate contaminants in saturated soil in situ. The electrokinetics/iron wall process appears to be a viable option in meeting the above challenges. The main strength of the electrokinetics/iron wall process lies in its ability to move water and dissolved contaminants(i.e. NO3--N, etc.) through low permeability soils by electroosmosis and electromigration to a location where they can be treated within the iron wall.
Electrokinetics is a process in which a low-intensity DC electrical current is applied across electrode pairs that have been implanted in the ground on each side of the contaminated soil mass. The process consists of several phenomena that result from the application of an electrical potential or current. Two principal phenomena of concern in this research are electroosmosis and electromigration. Electroosmosis is the phenomenon in which the pore fluid in the porous media moves when a potential or current is applied. The electroosmotic flow rate (q) is defined as follows: q = keieA; where ke is the coefficient of electroosmotic permeability; ie is the electrical potential gradient; and A is the cross-sectional area normal to the flow. Electromigration is the phenomenon in which solute ionic species are made to migrate to the anode or cathode depending on their respective charges. These include the H+ ions which are generated at the cathode and OH- ions that are generated at the anode due to the electrolysis of water. The H+ ions are more mobile than the OH- ions and therefore, move faster to the cathode than the OH- ions to the anode. This results in an acid front moving to the cathode and is likely to have an important effect in the electrokinetic process because speciation and solubility are often pH dependent.
In the iron wall, oxidative dissolution of Fe0 takes place resulting in the formation of Fe2+ ions. The redox couple formed by the zero-valent metal iron Fe0 and the dissolved Fe2+ ions (Fe0 Fe2+ + 2e-) has a standard reduction potential of 0.44V (Sawyer, et al., 1994). This makes Fe0 a reducing agent to many redox labile substances. For example, a likely nitrate-nitrogen transformation reaction under the influence of Fe0 is as follows: Fe0 Fe2+ + 2e-, E0 = 0.44 V; 2NO3- + 12H+ + 10e- N2(g) + 4H2O, E0 = 1.24 V; the combined reaction becomes: 5Fe0 + 2NO3- +12H+ 5Fe2+ + N2(g) + 6H2O, Erxn = 1.68 V. The combined reaction has a positive electrode potential of 1.68V and therefore, is likely to be thermodynamically favorable under most conditions.
Although considerable research has been conducted on either electrokinetics or Fe0-promoted treatment, very few studies were concentrated on electrokinetics coupled with Fe0 treatment (Ho, et al., 1995). More information is needed to develop protocols for remediation of nitrate-contaminated groundwater and soil by the processes with electrokinetics coupled with Fe0 treatment. The long-term goal of this research is to develop Fe0-promoted electrokinetic processes for in situ remediation of contaminants in saturated soil with low permeability. The objective of this paper is to present the preliminary results of nitrate transformation using electrokinetics/iron wall processes, including the effects of different voltages, current, and iron well on transformation of NO3--N in saturated artificial soil; and the preliminary determination of operational conditions and parameters under which the processes can achieve their optimal operational effect.
Methodology
Materials
Several bench scale reactors, consisting of 2.5-inch diameter by 7-inch length acrylic glass tubes with sampling and monitoring ports, were designed to simulate the remediation process.. The clay specimen(RC-32 air floated kaolin clay) was provided by Thiele Kaolin Co., GA. The fine sand specimen (Unimin Silica Sand by Unimin Corp., CT) was purchased in a local hardware store. The nitrate solution was made with lab grade potassium nitrate crystals. Graphite powder and rods (used as electrodes) were purchased from Midwest Graphite, IL. Industrial grade iron powder (80 mesh) was purchased from US Metal Co. Two small 20-watt DC power supply units, Hewlett Packard Model No. E3612A, were used to supply constant current or voltage to the reactors.
Batch Studies
Zero-valent Fe treatment of NO3--N contaminated soil was conducted in 150 mL glass beakers. In the control experiment, 15g of sand and 15g of clay were mixed with a 10 mL 100 mg/L NO3-N solution (no iron powder was used) in a 150 mL beaker. In another 150 mL beaker, 10g of the 80 mesh iron powder was added to a mixture of 10g sand, 10g clay, and 10 mls of a 100 mg/L NO3-N solution. These beakers were sealed with parafilm for two days and then allowed to air dry for another two days. Samples were obtained and analyzed for NO3--N, NO2--N, and NH3--N. as described in the Control Experiments section.
Electrokinetic Reactor Set-up
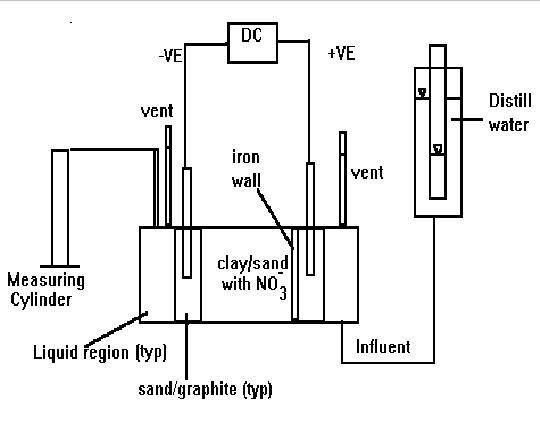
An artificially contaminated soil specimen (by w/w, 33% clay, 33% fine sand , and 33% of a 100 mg/L NO3--N solution) was loaded into a reactor cell and then sandwiched by an inch of sand and graphite mixture on both ends. The purpose of the sand/graphite layers was to help distribute the current evenly. A 20 g iron powder wall was placed near the anode to intercept nitrate ions migrating to the anode during the electrokinetic process. A small 20-watt DC power unit, with 3/4 inch graphite rods inserted into the sand/graphite layers, provided electricity to the cell. Water elevations at the influent and effluent were maintained at the same level to study the effects of electrokinetics and limit the hydraulic influences. Vents were provided near the cathode and anode to release any gases that may be generated during the process and thus prevent the hydraulic head from changing. Figure 1 shows a sketch of the bench unit constructed for the testing.
Control Experiments (electrokinetics only)
Three control runs were conducted using the setup shown in Figure 1 (except no iron wall was used) with constant voltages of 5, 10, and 15 volts. These constant voltages were supplied to each reactor for a period of 6 days. After 6 days, liquid samples were obtained from the liquid cathode and liquid anode regions. Soil samples were obtained from the sand/graphite regions and the contaminated soil region (sliced into five approximately equal parts) for analysis. The soil samples were air dried for 2-3 days and moisture contents were determined. Distilled water was added to the dried soils and thoroughly stirred. The slurried samples were then centrifuged at high speed with an International Clinical Centrifuge Model No. P6287. The supernates were used for analysis. All soil and liquid samples were analyzed for NO3--N, NO2--N, NH3-N and pH. NO3-N and NH3-N analyses were conducted using the ion-selective electrode method in accordance with the Standard Methods (APHA, et al., 1992). The NO2--N was determined colorimetrically with the HACH method (Hach Water Analysis Handbook, 1992) using a Milton Roy Spectronic 21 photometer. A mass balance was then performed to determine the percent of NO3--N lost after termination of the electrokinetic process. Also, pH of the soils was determined by mixing the dried soil samples with deionized water and measuring it with a combination type pH probe and meter.
Experiments of Electrokinetics Coupled with Fe0 Wall
A total of four runs at constant voltages of 5, 10, 15, and 20 volts and a total of two runs at constant currents of 1 and 2 mA were conducted to study the effects of voltage and current on the nitrate transformation process with 20g of iron powder placed near the anode to form an iron wall. The iron wall was placed near the anode so as to intercept the nitrate ions moving to the anode by electromigration. Samples were obtained and analyzed in the fashion similar to those for the control experiments.
pH Analysis
Three small tubes were inserted in the reactor along the artificially contaminated soil; one near the anode, one in the middle, and one near the cathode. These tubes allow pH readings to be taken along the contaminated soils on a daily basis to study the pH change in the three regions. These runs were conducted for a period of 9 days using both a control (no iron wall) reactor and a reactor with electrokinetics coupled with a 20g iron wall placed near the anode. pH measurements were made with an ISFET 100 pH probe/meter by Beckman.
Results
Batch Studies
In the control beaker, about 86% NO3--N by mass remained at the completion of the experiments. However, in the beaker where iron powder was used, only 2% of the NO3--N remained. Of the 98% NO3--N transformed, NH3-N accounted for 16%; NO2--N accounted for 1%; and the majority, i.e., 81%, was lost as some form of nitrogen gases.
Electrokinetic Control Runs (no iron wall)
Control runs were conducted to determine the effects of voltages on the nitrate-contaminated soil system using electrokinetic without any iron wall. For the mass balance, all unaccounted nitrogen after the soil and liquid sample analyses is assumed to be in the form of nitrogen gases. Figure 2 shows the forms of nitrogen remaining after the termination of the control experiments.
Total nitrate-nitrogen reduced ranged from 25% for 5 volts to 37% for 15 volts. Most of the nitrate transformed was in the form of nitrogen gases. Ammonia nitrogen accounted for less than 10% in all the runs. As shown in Figure 2, nitrate reductions increase with an increase in voltage supplied to the reactor. The average coefficient of electroosmotic permeability (Ke) values calculated for these runs was 2.56x10-6 (cm2/volt/sec) for 5 volts, 3.96x10-7 for 10 volts, and 4.06x10-6 for 15 volts, which were about one to two orders of magnitude lower than those reported by Mitchell (1976). However, these are average values (over a 6-day period) and there was little to no flow in the first day or two. These Ke values are not constant but changes with electrical conductivity (Acar, 1992) and therefore change as time progresses. The current through the reactor was negligible (due to the sensitivity of the power supply-minimum readout of 1 mA) for the 5- and 10-volt run. The current was about 1 mA in the 15-volt run but was negligible towards the end of the 6-day period. Flow was observed to increase when a current was registered on the meter. Moisture content in the soil was slightly less than the initial in the 5- and 10-volt run. However, in the 15-volt run, the moisture content ranged from 27% in the anode to 29 % in the cathode. This was due to soil consolidation (slight shrinkage was observed in the soil) during the electrokinetic process where water was driven to the cathode and the recharge rate from the influent was less than the water removal rate. The pH of the soils near the cathode (about 8.5) and anode (about 3) was consistent with the electrolysis of water process where H+ ions are generated at the anode and OH- ions in the cathode.
Constant Voltage with Fe0 Wall Experiments
These runs were conducted to study the effects of the different voltages and iron wall (in soil near the anode) on nitrate transformation. Figure 3 shows the forms of nitrogen remaining after the termination of the experiments. The iron wall placed in the reactor improved the NO3--N transformation. The amount of NO3--N reduced ranged from 54% for 5 volts to 87% for 20 volts under constant voltage conditions. As the transformation rate improved, more NH3-N was produced while the nitrogen gas produced remained relatively constant. Slight soil shrinkage and temperature increase were observed for the 20-volt run. Therefore, the constant voltage experiments were terminated after the 20-volt run. The current through the reactor was less than 1 mA for the 5-volt runs. The current was less than 1 to 2 mA in the 10- and 15-volt runs and 1 to 4 mA for the 20-volt runs. The average coefficient of electroosmotic permeability (Ke) values calculated for these runs was 2.86x10-6 (cm2/volt/sec) for 10 volts, 8.13x10-7 for 15 volts, and 4.33x10-6 for 20 volts. Since the current for the 5-volt runs was less than the 1 mA recording limitation, no calculation for the electroosmotic permeability coefficient was conducted. The shrinkage and temperature increased (0.5 0C) in the 20-volt run and may account for the slightly lower flow rates towards the end of the 6-day period. Acar (1993) reported that temperature decreases the electroosmotic flow. Also, as the ionic concentration in the pore water increases as a result of electrolysis, the electroosmotic flow is also reduced (Acar, 1993). In the constant voltage runs, the current decreased with time. This is because as the ionic concentration increases, electrical conductivity decreases (electrical conductivity is inversely proportional to voltage). pH analyses of the dried soils results were inconsistent with the expected results for the electrolysis process (about 3 - 4 in the anode and 8 - 9 in the cathode). The average power consumed was <13.5 (KwH/m3) for 5- and 10-volts runs, 13.5 for 15 volts, and 27 for 20 volts.
Constant Current with Fe0 Wall Experiments
The iron wall also improved the NO3--N transformation rate for the constant current runs.. Figure 4 shows the forms of nitrogen remaining after the termination of the experiments.
The transformation of NO3--N were 88% for the 1-mA runs and 84% for the 2-mA runs. As the transformation efficiency improved, more NH3-N was produced while the nitrogen gas produced decreased with an increase of ammonia production. The average coefficient of electroosmotic permeability (Ke) values calculated for these runs was 4.30x10-7 (cm2/volt/sec) for the 1-mA runs and 1.59x10-6 for the 2-mA runs. Final temperature in the soil increased slightly (10C) and substantial soil shrinkage was observed after 3 days of running the experiments for the 2-mA runs. As such, no further runs were made at constant current after the 1 and 2 mA runs. pH results appeared consistent with the expected results for the electrolysis process and were further evaluated in separate runs. In contrast to the constant voltage runs, the voltages increased with time for the constant current runs. The voltage range during the 6-day period was 6.6 to 15.2 volts for the 1-mA runs and 14-78 volts for the 2-mA runs. The average power consumed was <29.4 (KwH/m3) for the 1-mA runs and 124 for the 2-mA runs.
pH Analysis
Two reactors (one with iron wall and one without iron wall) were set up at a constant current of 1 mA to monitor the pH change with time. Three tubes were inserted in the port holes along the contaminated soil medium. Daily pH readings were taken for a period of 9 days in the tubes. The power was turned off to minimize to any electrical interference on the pH probe during the measurements. Figures 5a and 5b show time courses of pH profiles. In the control reactor (Figure 5a), the pH at cathode area increased from about 5.5 to 8.3 in the first 4 days and then decreased with time. At the end of the test (9 days), the cathode's pH was 5.5 again. The pH in the soil of the middle range of the reactor and near the anode decreased by 1 pH unit, indicating the movement of an acid front. However, this acid front did not flush across the cathode (pH remained high). In the iron wall reactor (Figure 5b), the results indicated that the pH near the anode increased with time, which was inconsistent with the phenomenon of pH increase in Fe0-water systems. The pH near the cathode area and in the soil of the middle range of the reactor decreased with time but the pH values were higher than those in the control reactor.
Discussions
The main strength of the electrokinetics lies in its ability to move water and dissolved contaminants (e.g. NO3--N in this study) through low permeability soils by electroosmosis and electromigration, which are not amenable to hydraulic in situ treatment wall and pump-and-treat strategies. However, unlike the treatment wall, the electrokinetics cannot remediate contaminants in situ. On the other hand, the success of a treatment wall strongly depends on characterization of the contaminant, groundwater flux, and subsurface geology. With an Fe0-promoted treatment wall between two electrodes, we can in situ-remediate contaminants which otherwise need to be collected at the electrode and then treated by conventional means. This study demonstrates that electrokinetics/iron treatment wall processes can be used efficiently for in situ remediation of nitrate-contaminated groundwater and subsurface soils in a saturated zone.
The results obtained from this study indicated that the process with electrokinetics/iron wall increased nitrate transformation efficiencies by 2 to 4 times, with highest removal efficiency of nitrate close to 90%. This high nitrate transformation efficiency with pH in the range of 4 to 10 (Figure 5b) is unusual for a Fe0-nitrate-water system where high nitrate removal can only be achieved at very low liquid pH (e.g., pH < 2) or with an addition of some organic buffer (e.g., HEPES) (Zawaideh, et al., 1997). The production of a harmful nitrite was always lower than the MCL (1 mg/L) set up by the U.S. EPA. The major transformation product was ammonium/ammonia, which may be transformed back to nitrate by nitrifying bacteria, a drawback of the process. However, according to Stumm and Morgan (1996), the distribution of different forms of nitrogen depends on the redox potential and pH in the system. It is possible to manipulate these two parameters to achieve higher transformation of nitrogen gas in the electrokinetics/iron well process. Therefore, an understanding of the process should be acquired to use the process effectively.
Summary
This research has indicated that the electrokinetic/iron wall process can be used to remediate nitrate-contaminated groundwater. Higher voltages and currents tend to consolidate the soil and can cause a slight temperature rise which can lower the flow rate . Two localized pH conditions exist in the system, a low pH region near the anode and a high pH region near the cathode. Placing of an iron wall near the anode increases the pH in that area as time increases. Movement of the acid front did not flush across the cathode. It is estimated that for about 75% NO3--N removal , approximately 13.5 KwH/M3 would be required. This would amount to about $1,350 of electrical charges (at $0.1/KwH) for removing nitrate in about 1000 M3 of soil.
Acknowledgments
Although this article has been funded in part by the U.S. Environmental Protection Agency under assistance agreement R-819653, through the Great Plains/Rocky Mountain Hazardous Substance Research Center headquartered at Kansas State University, it has not been subjected to the agency's peer and administrative review and therefore may not necessarily reflect the views of the agency, and no official endorsement should be inferred. The College of Engineering and Technology at the University of Nebraska-Lincoln (UNL) also partially funded this project. The Center for Infrastructure Research and the Water Center/Environment Programs of the UNL provided the matching funds for the project. During the whole study, Drs. P. Shea and S. Comfort in the Agronomy Department at UNL provided technical advice, which is greatly appreciated.
References
Acar, Y.B., Li, H., and Gale, R.J. (1992) Phenol removal from kaolinite by electrokinetics, Envir. Engrg., ASCE, 118:1837-1852.
Acar, Y.B., Alshawabkeh, A.N., and Gale, R.J. (1993) Fundamentals of extracting species from soils by electrokinetics, Waste Management, 13:141-151.
APHA., AWWA., and WEF (1992) Standard Methods for the Examination of Water and Wastewater. 18 th Ed.. American Public Health Association, American Water Works Association, and Water Environmental Federation, Washington, D.C.
Exner, M.E., and Spalding, R.F. (1990) Occurrence of pesticides and nitrate in Nebraska groundwater, Water Center Pub., No.1, Institute of Agriculture and Natural Resources, Univ. of Nebraska-Lincoln.
Hach Water Analysis Handbook. (1992). 2nd. Ed.. Hach Company, Loveland, Colorado.
Ho., S.V., Sheridan, P.W., Athmer, C.J., Heitkamp, M.A., Brackin, J.M., Weber, D., and Brodsky, P.H. (1995) Integrated in situ soil remediation technology: the lasagna process. Environ. Sci Tech., 29:2528-2534.
Mitchell, J.K. (1976) Fundamentals of soil behavior, John Wiley and Sons, New York, N.Y.
Spalding, R.F., and Exner, M.E. (1991) Nitrate contamination in the contiguous United States, NATO ASI series G 30, Nitrate Contamination, Ed. by Bogardi, I., and Kazelka, R.D., p.13-48, Springer-Verlag, Berlin
Stumm, W., and Morgan, J.J. (1996) Aquatic Chemistry, 3rd Ed., Wiley-Interscience, NY, 1996.
Zawaideh, L. L., Chew, C.F., and Zhang, T.C. (1997) Remediation of nitrate-contaminated water and soil by Fe0-promoted processes. 12th Annual
Conference on Hazardous Waste Research, May 19-22,Kansas City, MO, 1997.

Figure 1. Schematic diagram of a bench scale electrokinetic/iron wall reactor.
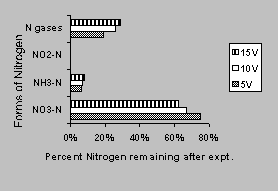
Figure 2. Nitrate transformation in control
runs at various voltages.
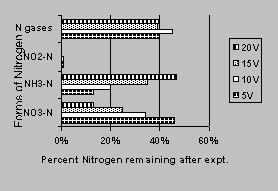
Figure 3. Nitrate transformation in reactors
with iron wall located
near the anode at various voltages
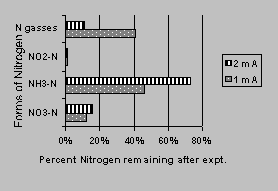
Figure 4. Nitrate transformation in reactors
with iron
wall located nearthe anode at various currents.
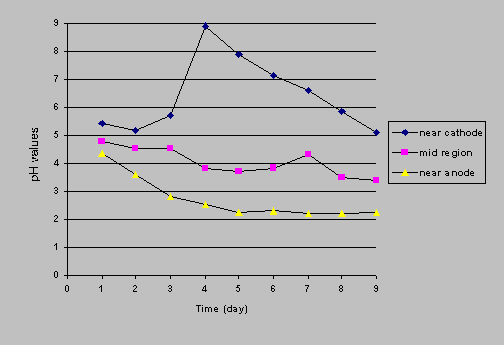
Figure
5a. Time courses of pH profiles (control reactor)
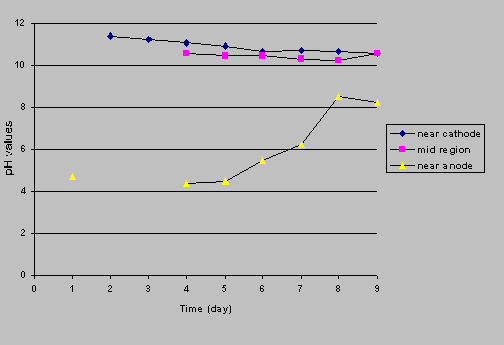 Figure
5b. Time courses of pH profiles (iron wall reactor)
Figure
5b. Time courses of pH profiles (iron wall reactor)